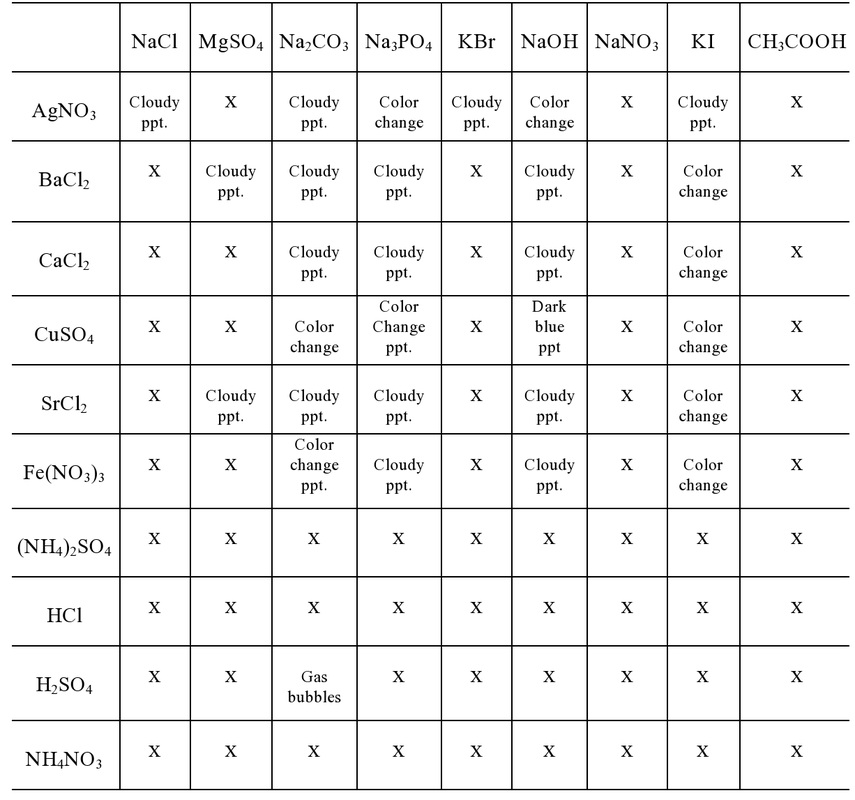
Data table:
Conclusion:
Based on the data from the experiment, general statements describing the solubility of specific cations and anions were made. All substances paired with phosphate (PO4 3-) is insoluble except Group 1 and ammonium. Ammonium sulfate, chloric acid, sulfuric acid, and ammonium nitrate did not form a precipitate. Therefore, when the reactants switch "partners" in the double replacement reaction ammonium and hydrogen determines whether the compound will be soluble or insoluble. All nitrates are soluble. In the experiment, of all the possible compounds nitrate could have formed, none of the solutions precipitated. All acetate (C2h3O2-) are soluble for the same reason all nitrates are soluble. Most substances paired with carbonate (CO3 2-) is insoluble, forming a solid. Carbonate formed a precipitate with most of the compounds except ammonium and HCl. However, it produce a gas with H2SO4.
Discussion of Theory:
Some of the reactant that were mixed together in the lab did not result in a precipitate, color change, or generation of a gas. This is due to the idea that "like dissolves like." When a nonpolar compound is mixed in a polar solution the mixture will form a precipitate because the polar solution cannot dissolve nonpolar compounds and vice versa. Polarity determines whether the solute will dissolve into the solvent.
Water is known as the universal solvent because of its polarity. Oxygen is more electronegative than hydrogen and it results in an unequally sharing of the electrons. It leave oxygen with a partially negative charge and hydrogen with a partially positive side. In the hydration of ionic solute molecules positive ions are attracted to the oxygen end of the water and negative ions are attracted to the hydrogen end of the water. In the hydration of polar solute molecules the negative ends of the polar solute molecules are attracted to the hydrogens (partially positive) and the positive ends of the polar solute molecule are attracted to the oxygen (partially negative).
A reaction does not react if the end products are aqueous. This is because the aqueous solutions that it began had the ions spilt up and at the end of the reaction the ions are still spilt up and floating around. The reactants remain the same and nothing new formed. Therefore, two aqueous products results in a no reaction. Depending on the strength of the electrolytes, some completely ionized when dissolved in water and others slightly ionize. Weak electrolytes are slightly ionized because the attraction to the water's partial charges are not as strong in weak electrolytes. When a substance is soluble it is a strong electrolyte because the concentration of ion in the solution is high because they were spilt apart by water. An electrolyte is a substance that can conduct electric currents, the flow of electrons. Weak electrolytes cannot conduct as much as strong electrolytes because the concentration of ions are higher in strong electrolytes. Therefore, more ions are available to move around in the solution.
A net ionic equation leaves the spectator ions out. Spectator ions are the ions that do not change at the end of the reaction. The complete ionic equation shows all of the ions that dissociate and the compounds that stay together (solids, liquids, & gases). Strong acids and bases alos dissociate and are written in ions. A net ionic equation shows exactly what reactants are involved in the reaction. It allows us to see what caused the solids, liquids, or gases to form.
A metathesis reaction is when cations and anions switch partners (double replacement reaction). The ions switch partners because opposites attract. The cations (positive) and anions (negative) are attracted to each other. The hydration of water, due to water polarity, helps keep the ions separated in the solution. However, when two aqueous solutions are mixed the anions and cations come together and form a precipitate, or gas. There are 3 types of metathesis reactions: precipitation reactions, neutralization reactions, and gas formation reactions. Most of the reactions in the experiment formed a precipitate. Those reactions are referred to as precipitation reactions. Neutralization reactions are the reactions that produces water and a salt. Neutralization reactions eliminate the acid (H+) and base (OH-) to form water as a product. By getting rid of the H+ and OH-, it neutralizes the reaction. Lastly, the solutions that gave off gas (bubbles) are referred to as gas formation reactions. For example, H2SO4 + Na2CO3 → Na2SO4 + H2O + CO2. Carbon dioxide gas was produced and bubbles were observed during the lab.
General statements about specific cations and anions were concluded because of the data from the experiment. The patterns that each cation or anion had contributed to the statement. When the double replacement reaction occurs the "partners" switch. The first ion in the compound determines whether a substance is insoluble or soluble. This is because most solubility rules have exceptions. The exceptions are based on the ions that they are paired with. Some ions do not have exceptions. For example, nitrates, acetates, ammonium and group 1 salts. Regardless of what these ions are paired with, they will always be aqueous.
Water is known as the universal solvent because of its polarity. Oxygen is more electronegative than hydrogen and it results in an unequally sharing of the electrons. It leave oxygen with a partially negative charge and hydrogen with a partially positive side. In the hydration of ionic solute molecules positive ions are attracted to the oxygen end of the water and negative ions are attracted to the hydrogen end of the water. In the hydration of polar solute molecules the negative ends of the polar solute molecules are attracted to the hydrogens (partially positive) and the positive ends of the polar solute molecule are attracted to the oxygen (partially negative).
A reaction does not react if the end products are aqueous. This is because the aqueous solutions that it began had the ions spilt up and at the end of the reaction the ions are still spilt up and floating around. The reactants remain the same and nothing new formed. Therefore, two aqueous products results in a no reaction. Depending on the strength of the electrolytes, some completely ionized when dissolved in water and others slightly ionize. Weak electrolytes are slightly ionized because the attraction to the water's partial charges are not as strong in weak electrolytes. When a substance is soluble it is a strong electrolyte because the concentration of ion in the solution is high because they were spilt apart by water. An electrolyte is a substance that can conduct electric currents, the flow of electrons. Weak electrolytes cannot conduct as much as strong electrolytes because the concentration of ions are higher in strong electrolytes. Therefore, more ions are available to move around in the solution.
A net ionic equation leaves the spectator ions out. Spectator ions are the ions that do not change at the end of the reaction. The complete ionic equation shows all of the ions that dissociate and the compounds that stay together (solids, liquids, & gases). Strong acids and bases alos dissociate and are written in ions. A net ionic equation shows exactly what reactants are involved in the reaction. It allows us to see what caused the solids, liquids, or gases to form.
A metathesis reaction is when cations and anions switch partners (double replacement reaction). The ions switch partners because opposites attract. The cations (positive) and anions (negative) are attracted to each other. The hydration of water, due to water polarity, helps keep the ions separated in the solution. However, when two aqueous solutions are mixed the anions and cations come together and form a precipitate, or gas. There are 3 types of metathesis reactions: precipitation reactions, neutralization reactions, and gas formation reactions. Most of the reactions in the experiment formed a precipitate. Those reactions are referred to as precipitation reactions. Neutralization reactions are the reactions that produces water and a salt. Neutralization reactions eliminate the acid (H+) and base (OH-) to form water as a product. By getting rid of the H+ and OH-, it neutralizes the reaction. Lastly, the solutions that gave off gas (bubbles) are referred to as gas formation reactions. For example, H2SO4 + Na2CO3 → Na2SO4 + H2O + CO2. Carbon dioxide gas was produced and bubbles were observed during the lab.
General statements about specific cations and anions were concluded because of the data from the experiment. The patterns that each cation or anion had contributed to the statement. When the double replacement reaction occurs the "partners" switch. The first ion in the compound determines whether a substance is insoluble or soluble. This is because most solubility rules have exceptions. The exceptions are based on the ions that they are paired with. Some ions do not have exceptions. For example, nitrates, acetates, ammonium and group 1 salts. Regardless of what these ions are paired with, they will always be aqueous.
Source of Error:
There were many areas where an error could have occurred in the lab. The results were recorded after all of the solutions were mixed together. This resulted in some of the solutions standing by for 10-15 minutes before recording the qualitative data. The solutions that released a gas could of had visible bubbles, but they were no longer there. There were many bottles with the different solutions labeled on the bottles. However, the experimenter could have mistakenly picked up the wrong solution and therefore, resulting in false data. The bottles of solution could have been contaminated prior to using them in lab. The systematic errors could be corrected by verifying with the equation. If the equation proves that a solid did form, but a murky/cloudy solution did not form it can be concluded that there was a systematic error. If the white cloudy precipitate was blending in with the white paper background, the error could easily be avoided by writing out the equation.