Purpose
The purpose of the lab is to experimentally calculate the concentration of NaOH using a titration with 10 mL of 1.5M HCl.
Procedure
1. Add 50 mL of an unknown concentration of NaOH to the buret. Record the starting volume for NaOH
2. Add 10 mL of 1.5M HCl to the Erlenmeyer Flask.
3. Add 2-3 drops of phenolphthalein to HCl.
4. Turn the stopcock to let the NaOH drip until the solution shows a faint tint of pink. The solution will turn pink at first, but the color will fade away after swirling the solution. As the solution approaches the equivalence point, the pink will take more time to fade away. Record the volume of NaOH on the buret in the ending volume column.
5. Repeat this process 3 more times. If the solution turn a hot pink in the first trial, it indicates the solution went slightly beyond the equivalence point.
6. Subtract starting volume from ending volume to get the amount of NaOH used.
7. Pour the solution in flask into the sink when finished. Be sure to wash the flask in between the trial and make sure it is completely dry before starting again.
2. Add 10 mL of 1.5M HCl to the Erlenmeyer Flask.
3. Add 2-3 drops of phenolphthalein to HCl.
4. Turn the stopcock to let the NaOH drip until the solution shows a faint tint of pink. The solution will turn pink at first, but the color will fade away after swirling the solution. As the solution approaches the equivalence point, the pink will take more time to fade away. Record the volume of NaOH on the buret in the ending volume column.
5. Repeat this process 3 more times. If the solution turn a hot pink in the first trial, it indicates the solution went slightly beyond the equivalence point.
6. Subtract starting volume from ending volume to get the amount of NaOH used.
7. Pour the solution in flask into the sink when finished. Be sure to wash the flask in between the trial and make sure it is completely dry before starting again.
Data Table
|
Trial
|
Starting volume (mL)
|
Ending volume (mL)
|
Change in volume (mL)
|
|
1
|
1.5 mL
|
13.2 mL
|
11.7 mL
|
|
2
|
13.20 mL
|
26.2 mL
|
13 mL
|
|
3
|
26.2 mL
|
37.8 mL
|
11.6 mL
|
|
4
|
37.8 mL
|
49.1 mL
|
11.3 mL
|
*Trials 1 & 2 resulted in a hot pink solution*
Calculations
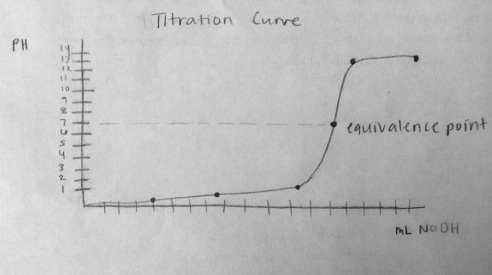
Graph
Conclusion
Based on the experimental data collected, the pH of NaOH is 1.26M. The titration formula was used to calculate the concentration.
Discussion of Theory
A titration is used to find the concentration of a particular solution with an unknown concentration by adding it to a solution of a known concentration. The solution with the known concentration is called the titrant. In order to visual see a color change, an indicator needs to be added to the solution. Indicators exist in solutions as HIn and In-. HIn and In- produce 2 different colors. The indicator used in the experiment, phenolphthalein, is clear if the solution is acidic and pink when the solution is basic. The color change occurs because if an acidic solution is added to the solution, more HIn is produced. If a basic solution is added, more In- is produced. For the experiment, the NaOH was added until the solution turned a faint pink for at least 30 seconds. Since more In- was produced, the solution turned faint pink. There must be a 10:1 ratio of the dominate form produced to change the solution color. This point is called the equivalence point. The equivalence point is where the number of moles of base equal the number of moles of acid. As the solution approaches the equivalence point, the indicator color will take longer to fade away. One extra drop could cause the solution to completely change because the endpoint and equivalence point are very close to each other. The end point is the point where the reaction is complete. Therefore, once the endpoint is hit the solution will not go clear (or the other direction). After the amount of NaOH need to neutralize HCl is obtained, the volume can be plugged into the titration equation. The titration equation is (M1V1)/n=(M2V2)n, where n= the mole to mole ratio. This is calculated by balancing the reaction. By plugging in the given and experimental data, the concentration of the unknown solution can be calculated. If a solution were to resist change, a buffer is required. A buffer is a solution that resists a change in pH when either hydroxide ions or protons are added. A buffer solution iis a solution containing common ions. They can either be a weak acid and its salt (conjugate base) or a weak base and its salt (conjugate acid).
Questions
1. What is the purpose of doing a titration?
The purpose of doing a titration is to find the concentration of a solution.
2. What is an indicator (in detail!)?
Indicators exist in solutions as HIn and In-. Each form has a distinct color. Depending on the solution containing the indicator, if an acidic solution is added to the solution, more HIn s produced. If a basic solution is added, more In- is produced. Whichever form is more dominate is the color that is expressed.n indicator is usually a weak acid or base. In order for a color change, the ratio of the strong acid/strong base and its conjugate base/acid must be 10:1. The color change occurs very rapidly at the equivalence point, the point is where the number of moles of base equal the number of moles of acid. As the solution approaches the equivalence point, the indicator color will take longer to fade away.
3. How do you decide which indicator should be used for a titration?
The indicator is selected based on the equivalence point. A good indicator will change colors at the equivalence point, so the indicator should change colors at the pH of the equivalence point of the solution.
4. What is the difference between the equivalence point and the end point?
The equivalence point is the point where the number of moles of base equal the number of moles of acid and the end point is the point where the reaction is complete. If the correct indicator is chosen it will change colors very close to the equivalence point. The endpoint and equivalence point are very close to each other. This explains why the color change can happen very quickly if a little over the equivalence point is dropped.
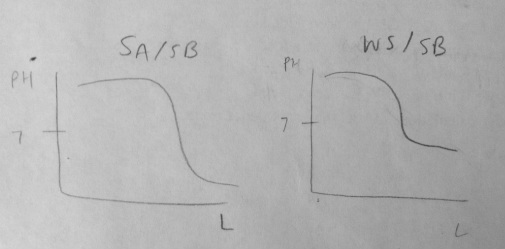
5. Compare and sketch a titration graph for a strong acid/strong base titration and for a weak acid/strong base titration.
Graph at the bottom of the page.
The strong acid/strong base drops to a lower pH unlike the weak acid/strong base titration. This is because the strong acid and strong base balance each other, however, the strong base is stronger than the weak acid so the solution is more basic.
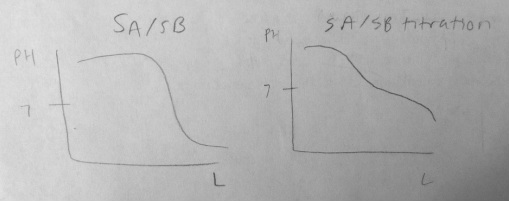
6. Compare and sketch a titration graph for a strong acid/strong base titration and the same titration after a buffer solution has been added.
Graph at the bottom of the page.
After a buffer has been added the graph has leveled off at the equivalence point.
7. Explain what a buffer is and how a buffer solution keeps the pH from changing.
A buffer prevents a solution from changing colors by adding H+ or OH-. These ions can be contributed by a salt which splits into its conjugate or a weak acid/base. LeChateliers Principle states that the reaction will shift to maintain equilibrium. The buffer reacts with additional H+ or OH- to resist change.
8. Consider each of the following potential sources of error. Answer:
“H” if it would have caused your calculated value for Molarity of NaOH to come out too high
“L” if it would have caused it to come out too low.
“N” if it would have no effect on your value.
a. There was a little distilled water in the Erlenmeyer flask before you began the titration.
(N) It would not affect the pH because liquids are not included in the equation. Water will only cause the HCl to ionize, which do not change the mole to mole ratio.
b. There was a little HCl in the Erlenmeyer flask before you began your titration.
(L) If there was a little HCl in the Erlenmeyer flask before you began your titration it would have cause the calculated value for molarity of NaOH to come out too high because the concentration of H+ is going to go up.
c. You added 3 drops of phenolphthalein, instead of 2 drops.
(N) Phenolphthalein is an indicator. It is not included in the equation, so it will not affect the molarity.
d. An air bubble was present in the NaOH buret, but it stayed in while you titrated.
(N) It would not affect it because you want the difference in the concentration. When you find the difference it will cancel out the volume of the air bubble.
e. An air bubble was present in the NaOH buret, and it came out in the middle of your titration.
(H) If there was an air bubble and it came out in the middle of your titration it will be accounted for. The volume we plug in will be higher than what the actual volume of NaOH used. It would be too high because the equation (M1V1)/n=(M2V2)/n multiplies volume and concentration. If the numerator is higher than it is suppose to be, then the calculated concentration of NaOH will be a higher.
f. While you were titrating, some NaOH dripped onto the table, instead of into the flask.
(H) The explanation is similar to the question above. The amount of NaOH actually used will be lower than the calculated difference on the buret. When you plug in a higher numerator, the overall value will be higher. Since we are solving for M2 in the equation, if all other number remain constant, the concentration will be too high.
g. You forgot to add the phenolphthalein indicator.
(N) Phenolphthalein is an indicator. It will not affect actual molarity of NaOH. The experimenter will not have been able to see a color change, so they would have to start over again. If they were to stop without judging based on a color change, then it will depend on where they stopped. If the volume was higher than the actual volume needed, then the calculated concentration would be too high. If the experimenter stopped at a volume lower than the actual volume needed, then the calculated concentration would be too low.
The purpose of doing a titration is to find the concentration of a solution.
2. What is an indicator (in detail!)?
Indicators exist in solutions as HIn and In-. Each form has a distinct color. Depending on the solution containing the indicator, if an acidic solution is added to the solution, more HIn s produced. If a basic solution is added, more In- is produced. Whichever form is more dominate is the color that is expressed.n indicator is usually a weak acid or base. In order for a color change, the ratio of the strong acid/strong base and its conjugate base/acid must be 10:1. The color change occurs very rapidly at the equivalence point, the point is where the number of moles of base equal the number of moles of acid. As the solution approaches the equivalence point, the indicator color will take longer to fade away.
3. How do you decide which indicator should be used for a titration?
The indicator is selected based on the equivalence point. A good indicator will change colors at the equivalence point, so the indicator should change colors at the pH of the equivalence point of the solution.
4. What is the difference between the equivalence point and the end point?
The equivalence point is the point where the number of moles of base equal the number of moles of acid and the end point is the point where the reaction is complete. If the correct indicator is chosen it will change colors very close to the equivalence point. The endpoint and equivalence point are very close to each other. This explains why the color change can happen very quickly if a little over the equivalence point is dropped.
5. Compare and sketch a titration graph for a strong acid/strong base titration and for a weak acid/strong base titration.
Graph at the bottom of the page.
The strong acid/strong base drops to a lower pH unlike the weak acid/strong base titration. This is because the strong acid and strong base balance each other, however, the strong base is stronger than the weak acid so the solution is more basic.
6. Compare and sketch a titration graph for a strong acid/strong base titration and the same titration after a buffer solution has been added.
Graph at the bottom of the page.
After a buffer has been added the graph has leveled off at the equivalence point.
7. Explain what a buffer is and how a buffer solution keeps the pH from changing.
A buffer prevents a solution from changing colors by adding H+ or OH-. These ions can be contributed by a salt which splits into its conjugate or a weak acid/base. LeChateliers Principle states that the reaction will shift to maintain equilibrium. The buffer reacts with additional H+ or OH- to resist change.
8. Consider each of the following potential sources of error. Answer:
“H” if it would have caused your calculated value for Molarity of NaOH to come out too high
“L” if it would have caused it to come out too low.
“N” if it would have no effect on your value.
a. There was a little distilled water in the Erlenmeyer flask before you began the titration.
(N) It would not affect the pH because liquids are not included in the equation. Water will only cause the HCl to ionize, which do not change the mole to mole ratio.
b. There was a little HCl in the Erlenmeyer flask before you began your titration.
(L) If there was a little HCl in the Erlenmeyer flask before you began your titration it would have cause the calculated value for molarity of NaOH to come out too high because the concentration of H+ is going to go up.
c. You added 3 drops of phenolphthalein, instead of 2 drops.
(N) Phenolphthalein is an indicator. It is not included in the equation, so it will not affect the molarity.
d. An air bubble was present in the NaOH buret, but it stayed in while you titrated.
(N) It would not affect it because you want the difference in the concentration. When you find the difference it will cancel out the volume of the air bubble.
e. An air bubble was present in the NaOH buret, and it came out in the middle of your titration.
(H) If there was an air bubble and it came out in the middle of your titration it will be accounted for. The volume we plug in will be higher than what the actual volume of NaOH used. It would be too high because the equation (M1V1)/n=(M2V2)/n multiplies volume and concentration. If the numerator is higher than it is suppose to be, then the calculated concentration of NaOH will be a higher.
f. While you were titrating, some NaOH dripped onto the table, instead of into the flask.
(H) The explanation is similar to the question above. The amount of NaOH actually used will be lower than the calculated difference on the buret. When you plug in a higher numerator, the overall value will be higher. Since we are solving for M2 in the equation, if all other number remain constant, the concentration will be too high.
g. You forgot to add the phenolphthalein indicator.
(N) Phenolphthalein is an indicator. It will not affect actual molarity of NaOH. The experimenter will not have been able to see a color change, so they would have to start over again. If they were to stop without judging based on a color change, then it will depend on where they stopped. If the volume was higher than the actual volume needed, then the calculated concentration would be too high. If the experimenter stopped at a volume lower than the actual volume needed, then the calculated concentration would be too low.